BIOMEDICAL MULTI-MODAL ULTRASOUND IMAGING
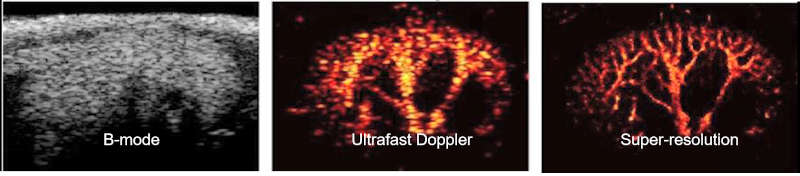
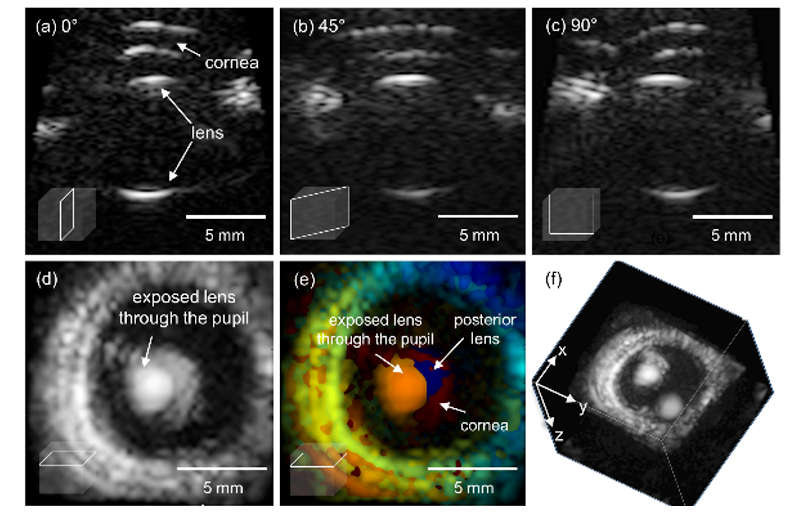
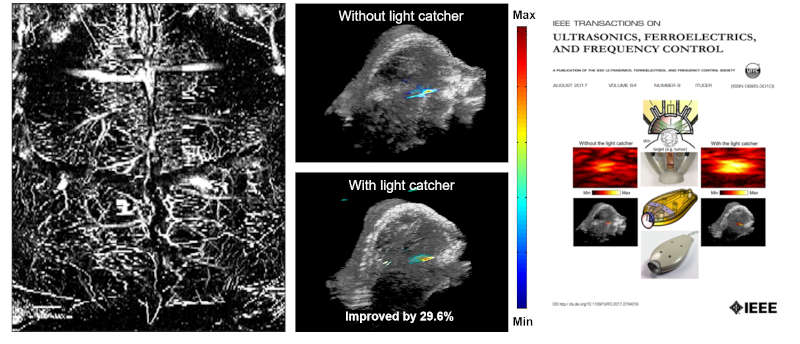
To provide a comprehensive understanding of complex diseases like cancer and cardiovascular conditions, we develop multi-modal ultrasound platforms. By integrating traditional B-mode sonography with advanced functional techniques—such as elastography (for tissue stiffness), thermal strain imaging (for composition), Doppler imaging (for hemodynamics), and contrast-enhanced imaging (for blood perfusion)—we can capture anatomical, mechanical, compositional, and optical information from a single imaging plane, simultaneously.
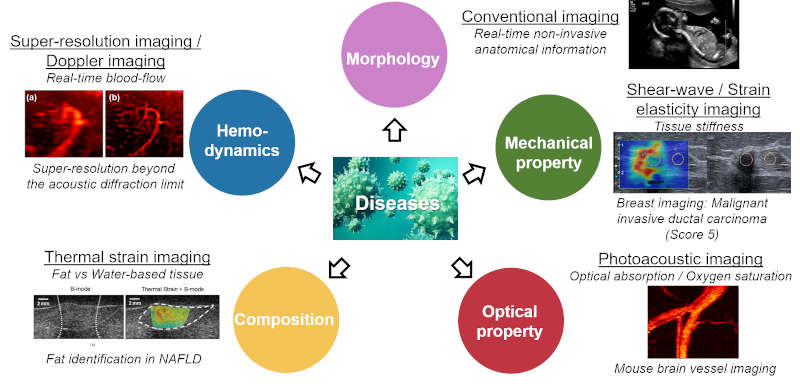
FIG. Multi-modal ultrasound imaging technologies